Latest News
17-Feb-2025GPHF-Minilab™: Application possibilities extended once again
The Global Pharma Health Fund (GPHF) has now further extended the range of applications of its Minilab for the identification of counterfeit or low-quality drugs. A total of 125 active pharmaceutical ingredients (APIs) can now be identified and analyzed with the mobile laboratory. There is also a special test for excipients to detect dangerous antifreeze as a toxic contaminant in children's syrups.
In order to optimize the market surveillance possible with the Minilab, which serves to protect potentially endangered patients, the six active ingredients diphenhydramine, hyoscine, loperamide, loratadine, norfloxacin and tinidazole have now been added to the laboratory's test methodology. The new methods are available for free download here in a manual in English, French and Spanish:
https://www.gphf.org/en/minilab/manuals.htm.
06-Feb-2025
GPHF-Minlab™ again proven in study
Counterfeit or substandard medicines are jeopardizing healthcare worldwide. A study conducted with the GPHF-Minilab, the results of which have now been published in the International Journal of Pharmacy and Chemistry, specifically examined the quality of antimalarial drugs containing the active ingredient combination artemether and lumefantrine that are available in Côte d'Ivoire. The results are alarming: of the 15 samples examined, 20% lacked the manufacturer's instructions, 7% were missing the instructions for use and 20% showed physical degradation. While 93% of the samples met the dissolution standards, 26% were underdosed and 7% were overdosed, suggesting manufacturing errors. According to the scientists who conducted the study, the GPHF-Minilab® proved to be an effective and reliable tool for identifying substandard and dangerous drugs on the market.
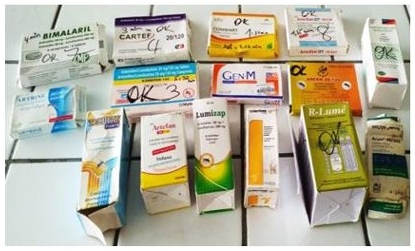
The drugs tested in Côte d'Ivoire.
10-Jan-2025
Project work successfully continued in 2024
The GPHF continued its successful project work in 2024. Last year, a total of seven additional active ingredients were added to the GPHF-Minilab's test methodology, which is now available for 119 individual active ingredients from a wide range of indication areas. In addition, 12 further laboratory units were delivered for the identification of counterfeit or poor-quality drugs and two training courses on the use of the mobile test laboratory were held in the South Pacific in Tonga and Nauru. Detailed information on the current status of the Minilab project (areas of application, active ingredients, manuals, etc.) can be found in the chapter “The GPHF Minilab”.
13-Dec-2024
New study once again proves the benefits of the GPHF-Minilab™
Low-quality or counterfeit medicines are a serious threat to public health care, especially in low- and middle-income countries. The visual inspection of medicines and screening analyses using the Minilab developed by the Global Pharma Health Fund to monitor the quality of medicines have proven their worth, particularly in resource-poor countries and regions. This has now been proven again by a study in Nigeria. You can find the details here.

Figure from the study results published in the Journal of Pharmaceutical Policy and Practice.
28-Nov-2024
Fight the Fakes Week 2024
This year's campaign week of the international Fight the Fakes initiative will take place from December 2 to 8. This year's focus is the white paper “Identifying Critical Enablers in the Prevention, Detection and Response to Substandard and Falsified Medicines”, which will also be discussed in a special webinar on December 5.
Fight the Fakes is an international initiative that draws worldwide attention to the dangers of substandard and falsified medicines and is also supported by the Global Pharma Health Fund e. V. (GPHF). Further information on this year's campaign week, the white paper and the webinar can be found here.

20-Nov-2024
GPHF-Minilab™ again proven in practice
A scientific study has now once again underlined the value of the GPHF-Minilab™ for the control and safety of medicines - and the quality of the resulting care for patients. In Ghana, it was investigated how different methods of determining the quality of medicines can work together to ensure safe patient care. The result: both the minilab and digital screening methods can make an important contribution to this. You can find more information here.
14-Oct-2024
Warning about toxic antifreeze
The World Health Organization has once again drawn attention to the current risk of contamination of medicines by toxic antifreeze. Specifically, it concerns a case from Pakistan, where batches of contaminated raw materials for raw materials for the production of medicines were discovered. The corresponding warning can be found here.
Just recently, the Global Pharma Health Fund (GPHF) conducted a test as part of its Minilab project, the Global Pharma Health Fund (GPHF) conducted a test to detect the toxic antifreeze diethylene glycol (DEG) and ethylene glycol (EG). In the past, these were repeatedly found in cough syrups for children, which then pose a fatal danger.
All information on the new GPHF test can be downloaded here free of charge in English, French and Spanish.
04-Sep-2024
GPHF-Minilab: New test for the detection of toxic antifreeze agents
As part of its Minilab project to combat counterfeit and substandard medicines, the Global Pharma Health Fund (GPHF) is now providing a test to detect the toxic antifreeze agents diethylene glycol (DEG) and ethylene glycol (EG).
The toxic industrial solvents DEG and EG have been repeatedly found in children's cough syrups in the past and pose a fatal risk as they can lead to kidney failure, especially in children. The new test is intended to support national quality control laboratories in particular to detect DEG/EG contamination and prevent the use of adulterated liquid preparations for oral use.
The new test protocol can be downloaded here free of charge in English, French and Spanish and includes a QR code to access a complementary video tutorial.
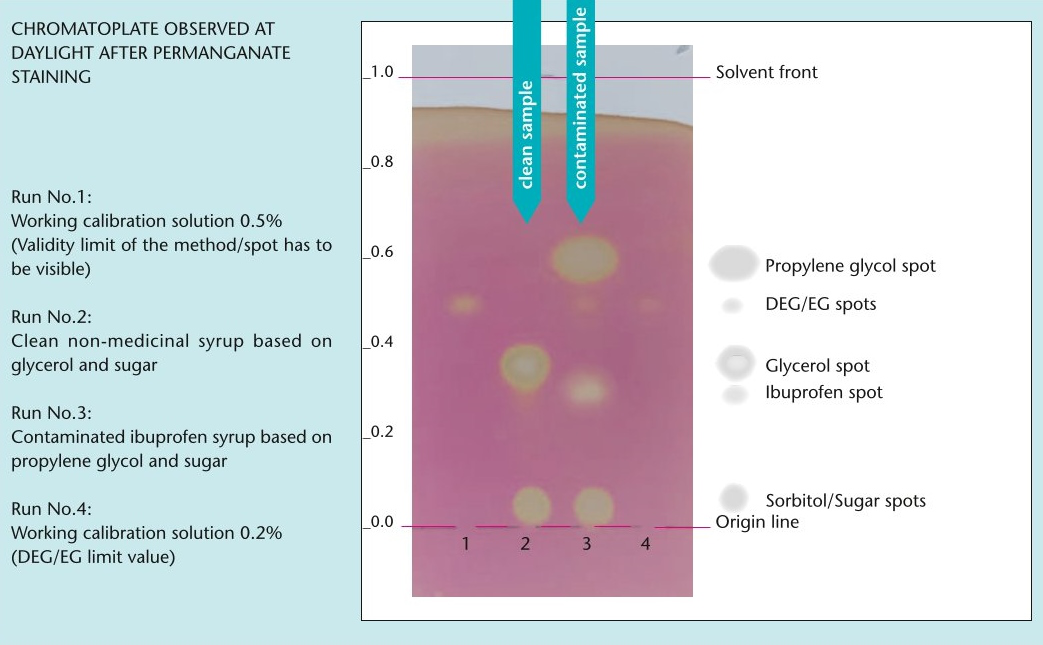
Excerpt from the test protocol.
17-May-2024
GPHF-Minilab™ presented in the Bundestag
The Chairman of the GPHF, Dr. Johannes Waltz, recently had the opportunity to present the GPHF's commitment to combating counterfeit drugs and the GPHF-Minilab developed by the GPHF for this purpose to the Health Committee of the German Bundestag in Berlin.
The Subcommittee on Global Health, which is chaired by Professor Dr. Andrew Ullman (FDP), had put the issue of counterfeit drugs on the agenda and invited various organizations, including the GPHF, as experts.
Afterwards, the GPHF Chairman was very impressed by the MPs' interest in the GPHF's experience and expressed the hope that this would also raise awareness of the GPHF's mobile compact laboratory among political decision-makers.
The Global Health Subcommittee was set up during this legislative period within the Health Committee and deals specifically with international health issues. This includes the issue of counterfeit medicines, which has long been a global challenge for healthcare systems, affecting countries in Africa and South East Asia in particular.
13-May-2024
Minilab training courses in the South Pacific
At the invitation of the World Health Organization, the GPHF has now conducted two one-week training courses for the use of the GPHF Minilab in the South Pacific. GPHF project manager Dr. Richard Jähnke successfully prepared a total of 14 pharmacists on both Tonga and Nauru for their future work with the mobile test laboratory for the identification of counterfeit or substandard medicines.
Counterfeit medicines have also long since become a serious health problem in the Pacific region. The use of the minilab is therefore intended to help improve the local control of medicines and increase the safety of the supply of medicines to the population. Thanks to the commitment of the World Health Organization, the GPHF mobile laboratories are already available in Fiji, Tonga, Nauru and Samoa, and there are plans to support more of the 21 island states in the region with GPHF Minilabs and corresponding user training in the future.

Dr. Richard Jähnke from the GPHF (center in the background) with the participants of the training course on Nauru.
19-Apr-2024
WHO warns of contaminated propylene glycol
Following investigations by the Pakistani drug authority, the World Health Organization has issued a warning about contaminated propylene glycol used in the production of medicines. As propylene glycol is one of the basic ingredients used in the production of syrup, it is often used in medicines for children and therefore poses particular dangers for them in the event of contamination, which can even lead to death. The contamination is usually due to ethylene or diethylene glycol.
The Global Pharma Health Fund (GPHF) is confident that it will soon be able to supplement its Minilab with a fast and cost-effective test protocol for detecting diethylene/ethylene glycol contamination in syrup so that this risk can be countered in the future.
The original World Health Organization document can be found here.
12-Mar-2024
GPHF-Minilab™ also in use in Mali
Malaria is also one of the greatest threats to human health in the West African country of Mali - affecting mothers, newborns and children in particular. This makes it all the more reprehensible when medicines used to treat the disease are of inferior quality or even counterfeit. In order to investigate the situation on the ground, samples of locally available medicines have now been taken in various regions of the country and tested using the GPHF's Minilab. The result: up to six percent of the medicines were substandard or counterfeit. As a result, the health authorities in Mali have taken various steps to improve the situation and protect the population. Read the full story here.

Pharmacists at a stocked pharmacy in Mali
15-Feb-2024
Further expansion of the Minilab's range of applications
With the inclusion of six additional active pharmaceutical ingredients, the GPHF has once again expanded the range of applications of its mobile laboratory for the detection of counterfeit drugs. A total of 119 active pharmaceutical ingredients can now be identified and analyzed with the GPHF-Minilab.
The six new test methods are all aimed at active ingredients for the treatment of diabetes, which is now also a highly relevant health problem in many countries of the global South. As counterfeit diabetes drugs have also emerged in the past, the GPHF hopes that by expanding the methodology of its Minilab, it will be able to contribute to the protection of patients in this area as well.
The description of the new Minilab methods is now available to download free of charge in English, French and Spanish here.
24-Nov-2023
Fight The Fakes Action Week 2023
The Fight The Fakes Alliance is holding its sixth annual week of action from December 4 to 10. This year's motto is "Africa Together Against Substandard And Falsified Medicines". The focus is on Africa, as the continent and its people are affected by the dangers of counterfeit and substandard medicines like no other. It is estimated that up to 500,000 people die every year south of the Sahara alone as a result of taking these types of "medicines".
The Fight The Fakes Alliance is an association of international organizations and is also supported by the Global Pharma Health Fund e.V. (GPHF). The aim is to raise awareness worldwide of the health risks posed by low-quality and counterfeit medicines and to protect people from the associated health risks.
Further information on this year's campaign week can be found here.
17-Oct-2023
Important building block for protection against counterfeit drugs
On the occasion of 25 years of the GPHF-Minilab®, international experts discussed the significance of the mobile laboratory in the context of the fight against counterfeit or low-quality drugs at this year's World Health Summit in Berlin under the chairmanship of Professor Lutz Heide (Pharmaceutical Institute of the University of Tübingen).
On behalf of the GPHF, Dr. Jutta Reinhard-Rupp pointed out by way of introduction that the GPHF was well aware of the fact that the mobile laboratory would not be able to solve the problem of counterfeit drugs on its own, because the challenge was simply far too complex for that. However, thanks to its special features - mobile, easy to handle, reliable and cost-effective - the minilab has proven to be an important element of drug control in many places. In the meantime, more than 1,000 laboratory units are already in use worldwide, which, according to the GPHF board member, make an important contribution to protecting people from a potentially lethal hazard.

Dr. Jutta-Reinhard-Rupp (center) referred to the important contribution made by the GPHF-Minilab to the protection against counterfeit drugs worldwide.
Pernette Bourdillon-Esteve from the World Health Organization in Geneva then made it clear that counterfeit medicines can completely undermine and destroy people's trust in the healthcare system and its efficiency. In order to prevent this in a targeted manner, he said, it is essential for the WHO to record the situation through monitoring that is as complete as possible. Only if we know where counterfeit medicines are appearing can we issue timely warnings and take targeted countermeasures, the expert said.
Finally, Richard Neci, Executive Director of the Ecumenical Pharmaceutical Network (EPN) in Nairobi, Stephen Kigera, Mission for Essential Drugs and Supplies (Kenya), Joanita Namutebi Lwanyanga, Joint Medical Store (Uganda), and Julia Gabel of the Pharmaceutical Institute of the University of Tübingen, who has carried out a research project in Nigeria on the survey of drug quality with the aid of the minilab, reported on their practical experience with the GPHF minilab. It became clear in the individual speeches not only that counterfeit or low-quality drugs continue to be a major problem in the countries mentioned, but also that the GPHF minilab makes it possible to quickly and reliably identify counterfeits as such.
In conclusion, the GPHF stated that it will continue to integrate new active pharmaceutical ingredients into the testing methodology of its mobile laboratory, and that the current number of 112 will thus be increased to 125 by 2025. However, he said that he is also open to technical innovations that may complement the laboratory's current testing methodology in the future.
18-Aug-2023
Minilab Training in Rwanda

In the East African country of Rwanda, a five-day introductory course has now been held on the future use of the GPHF mobile laboratory for the identification of counterfeit or low-quality drugs. The picture shows the course participants in front of the training center.
31-Jul-2023
Warning about contaminated cold syrup
The World Health Organization (WHO) is currently warning about the health hazards of a contaminated syrup sold under the trade name NATURCOLD, which is supposed to be used to relieve symptoms of colds, flu or allergic rhinitis.
The syrup was discovered in Cameroon in March of this year and interim analyses have revealed that the product contains unacceptable levels of diethylene glycol in addition to the declared active ingredients paracetamol, phenylephrine hydrochloride and chlorpheniramine maleate. In samples from NATURCOLD, diethylene glycol was detected in amounts up to 28.6%. However, the permissible limit is no more than 0.10%. In the past, cold or cough syrups contaminated with diethylene glycol have led to many deaths.
The company FRAKEN INTERNATIONAL from England is listed as the manufacturer of NATURCOLD. However, the national regulatory authority in the UK has stated that there is no such manufacturer in the UK. Investigations are currently underway to determine the origin of the product. More details on the WHO warning can be found here.
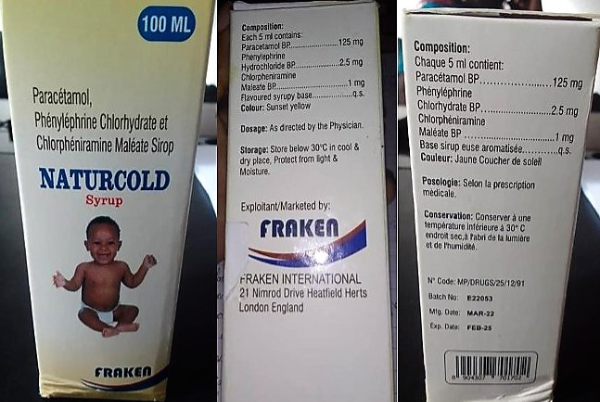
The contaminated cold syrup discovered in Cameroon. (Image: WHO)
30-Jun-2023
A special anniversary: The 1,000th minilab was delivered
It is a milestone for the Global Pharma Health Fund e.V. (GPHF), as the 1,000th GPHF Minilab has been delivered these days. The mobile compact laboratory, which bears the number 1,000 and which can be used to detect counterfeit or low-quality drugs, is part of an order for a total of 16 laboratory units which will be used in the future in the countries of the East African Union in order to protect the people there from what is often a deadly danger. Initially, the National University of Rwanda will be in charge of the local deployment and will conduct a training course for the future users of the labs. The 16 minilabs were financed with funds from the Kreditanstalt für Wiederaufbau (KfW) in Germany.
The concept of the minilab has meanwhile proved its worth for more than two decades, in about 100 countries and on all continents. Dr. Richard Jähnke of the GPHF, who designed the laboratory and has already used it in numerous countries: "Our method is simple, reliable and inexpensive. It enables the personnel in hospitals, health stations or similar institutions to examine the quality of the drugs available on site within a short period of time. In many places, this is a quantum leap, because such possibilities often did not exist at all before, and one can imagine what this may mean for patients."
In many cases, the GPHF-Minilab has already been able to detect and remove from circulation counterfeit drugs, which are particularly widespread in Africa and Southeast Asia. However, Dr. Jähnke and the GPHF are constantly working on further extending the possible applications of the minilab, i.e. to include additional active pharmaceutical ingredients in the methodology. At present, 113 important active substances for the treatment of a wide variety of diseases can already be identified and analyzed, and planning for further active substances has already started.
05-Apr-2023
Further active ingredients added to the GPHF-Minilab™ method inventory
The GPHF-Minilab now allows the identification of six additional active substances, the majority of which belong to the group of blood thinners and are used for the treatment of cardiovascular diseases. These are the active ingredients apixaban, candesartan, clopidogrel, hydralazine, rivaroxaban and warfarin.
Although cardiovascular diseases are among the leading causes of death worldwide, comparatively little attention has been paid to the counterfeiting of cardiovascular drugs. However, to also facilitate their monitoring in the supply chains of low- and middle-income countries, the Minilab's inventory, including the new methods, now includes twenty such agents. In total, the GPHF mobile laboratory can thus now identify 113 active pharmaceutical ingredients in various formulations for the treatment of communicable and non-communicable diseases.
The six new methods are available for free download here in English, French and Spanish.
20-Mar-2023
Counterfeit drugs: UNDOC report with shocking facts
A report now published by the United Nations Office on Drugs and Crime (UNDOC) comes to shocking conclusions. According to the report, in the sub-Saharan part of the African continent alone, up to 500,000 people fall victim to counterfeit or low-quality medicines every year. Those affected are mainly antibiotics and drugs to protect against malaria. This not only causes untold human suffering, but also immense economic damage. Further information on the report can be found here.
15-Dec-2022
GPHF General Meeting
Despite the difficult general conditions caused by the Corona pandemic, the Global Pharma Health Fund e.V. (GPHF) has now been able to draw a positive balance of its project work of the past three years at its general meeting. In the meantime, the number of active pharmaceutical ingredients that can be identified with the GPHF-Minilab™ has not only been increased to 107, but numerous additional laboratory units have also been ordered by international partners, so that the total number has now already risen to more than 960. The GPHF also regards it as a very positive signal that the German Association of Research-based Pharmaceutical Companies has become a member of the GPHF and has also declared its willingness to specifically promote the further technological development of the mobile test laboratory for the identification of counterfeit or low-quality drugs in the future.
In the course of the board elections, Dr. Johannes Waltz and Dr. Jutta Reinhardt-Rupp were confirmed in their positions as chairman and deputy chairman, respectively. Birgit Kröling-Neumann will also join the GPHF board as treasurer.
08-Dec-2022
Minilab Training on the Fiji Islands
For the first time since the beginning of the corona pandemic, the GPHF has now been able to conduct another minilab training course. At the invitation of the World Health Organisation (WHO), Dr Richard Jähnke of the GPHF travelled to the Fiji Islands at the end of November and instructed 15 staff members of the national health authorities and local hospitals at the WHO training center in Nadi in the handling of the mobile test laboratory for the identification of counterfeit or qualitatively inferior drugs. According to the GPHF, the one-week training course was very successful, as the participants, most of them pharmacists, were well qualified and highly motivated.
The WHO Regional Office for the Western Pacific, based in Manila, Philippines, had procured and distributed five GPHF minilabs for its Department of Pacific Technical Support (DPS) in Suva, Fiji, in late 2019, just before the start of the Corona pandemic. Five Pacific island countries (Fiji, Tonga, Nauru, Tuvalu, and the Solomon Islands) each subsequently received a GPHF minilab to strengthen drug safety in the field.

Impressions from the Minilab training in Fiji
28-Nov-2022
Fight The Fakes action week 2022
For the fifth time, the international action week of the Fight The Fakes initiative will take place from December 5 to 11. This year's theme is "Keeping Patents Safe By Securing The Global Medicines Supply Chain" and aims to highlight the importance of a safe and reliable supply chain for the global supply of medicines and patient safety.
The Fight The Fakes initiative is a coalition of international organizations and is also supported by the Global Pharma Health Fund e.V. (GPHF). Its aim is to draw worldwide attention to the health hazards posed by low-quality and counterfeit medicines and to protect people from the associated risks to their health.
Further information on this year's campaign week can be found here:
https://fightthefakes.org/week/5th-fight-the-fakes-week/
08-Aug-2022
The GPHF Minilab™ in Practice - A Systematic Observation
Under the title "Surveillance for substandard and falsified medicines by local faith-based organizations in 13 low- and middle-income countries using the GPHF Minilab", a study has now been published which for the first time documents, analyses and evaluates the use of the mobile test laboratory for the identification of falsified and low-quality medicines in larger geographical contexts - thirteen countries on two continents - and over a longer period of time - two years. The compact laboratory is proving to be a reliable, effective and cost-efficient method of monitoring drug quality in the field. The 13-page report of the study can be found here.
26-Jul-2022
USD 11 million in illicit medicines seized in global INTERPOL operation
Every day, advertisements for medicines invade the Internet, posted on social media networks or other websites. However, behind this slick marketing often lies fraudulent products that threaten consumers’ health instead of healing them.
The global trade in illicit pharmaceuticals is a vast and lucrative crime area – valued at USD 4.4 billion – which attracts the involvement of organized crime groups around the world.
Over just one week (23-30 June), 94 INTERPOL member countries representing every continent launched a coordinated crackdown on illicit online pharmacies in Operation Pangea XV. Globally, law enforcement made more than 7,800 seizures of illicit and misbranded medicines and healthcare products, totaling more than 3 million individual units. More information and many photos of the operation can be found here.

Illicit medical products uncovered by law enforcement in Panama. (Picture: INTERPOL)



