2019: Gefälschte Blutdrucktabletten in Kamerun enthalten Antidiabetikum
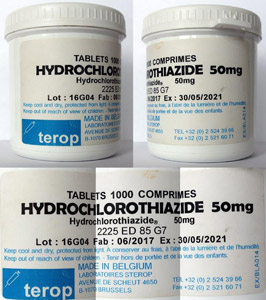
A nongovernmental organisation in Cameroon informed the local health authorities that a medicine presenting as hydrochlorothiazide 50mg had caused hypoglycaemia in patients. Preliminary Minilab testing indicated that the product did not contain any of the stated active ingredient, hydrochlorothiazide, and glibenclamide had instead been identified. Both results were later confirmed by fully-fledged lab testing. In addition, the batch number and expiry date displayed did not match the records of the company stated on the label. The replacement of a usual hydrochlorothiazide treatment course by a full glibenclamide dosage regime has serious consequences for patients. If glibenclamide is taken by non-diabetic patients, i.e. patients without elevated blood sugar levels, and/or by patients who are not informed about the detection and treatment of symptoms of hypoglycaemia, it can lead to hypoglycaemic shock and even death, both preventable by the intake of a spoonful of sugar. More on this case can be found at the Medical Product Alert system of the World Health Organization (WHO) and on GPHFs Facebook timeline.
« back
