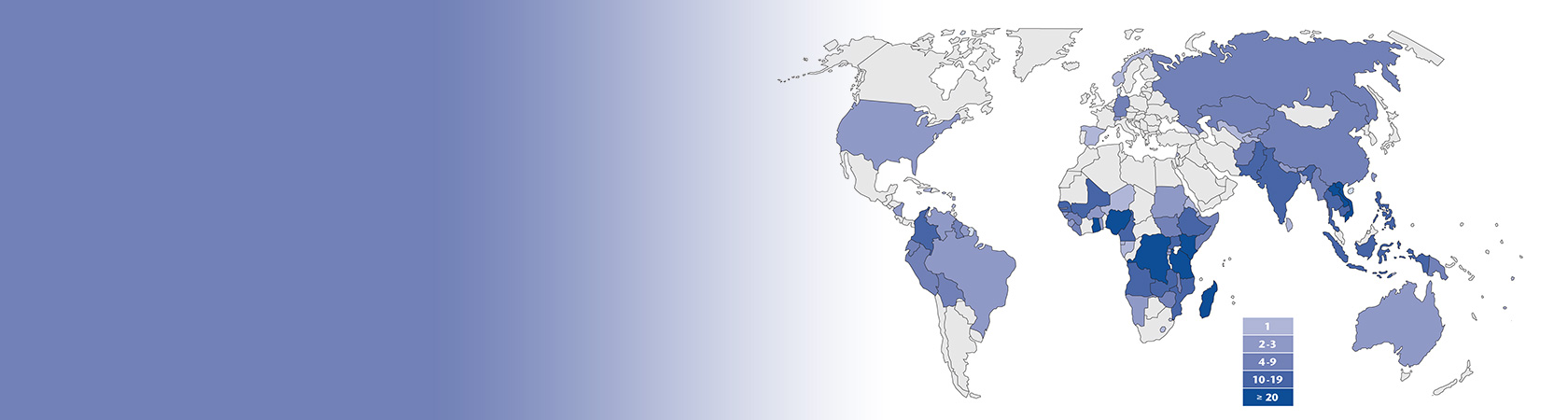
The GPHF-Minilab™ - Protection Against Counterfeit Medicines
 Due to an increase in the proliferation of fake and dodgy medicines and the individual and collective health risks associated with this phenomenon, drug quality control in the distribution systems of developing countries has become more and more important for consumer protection today. According to the latest research from the World Health Organization (WHO), 1 in 10 medical products circulating in developing countries is either substandard or falsified.
Due to an increase in the proliferation of fake and dodgy medicines and the individual and collective health risks associated with this phenomenon, drug quality control in the distribution systems of developing countries has become more and more important for consumer protection today. According to the latest research from the World Health Organization (WHO), 1 in 10 medical products circulating in developing countries is either substandard or falsified.
Under these circumstances, a greater number of samples have to be tested in order to maintain an appropriate assurance of drug quality and a high level of patient protection. Yet at the same time, pharmacopoeial analyses have become more and more expensive and only a few centers of excellence in some countries are currently available to perform them in the quantities required.
In order to bridge the capacity gap in regular drug quality monitoring on national level in low-income countries and in order to overcome limited access to regular drug quality testing of public, private and faith-based drug supplies, the Global Pharma Health Fund (GPHF) set out to develop and supply inexpensive field test kits with simple test methods for rapid drug quality verification and counterfeit medicines detection.
Rapid Counterfeit Medicines Detection anywhere in the World
 The GPHF-Minilab takes the basic drug testing scheme published by the World Health Organization (WHO) some thirty years ago into the 21st century. New test methods have been introduced, and supplied are not only operation manuals printed in different languages, but also a complete range of lab ware, starter kit chemicals and reference standards is included - all suitably packed for global shipment by air. Now, identification of counterfeit medicines containing wrong, too little, or no ingredients at all can be performed instantly anywhere in the world. Results obtained by a set of physical and chemical screening tests must match the product label claims for, at least, drug identity and content. If they do not match or results are inconclusive, then the appropriate batches can be frozen for further investigation in a fully-fledged lab.
The GPHF-Minilab takes the basic drug testing scheme published by the World Health Organization (WHO) some thirty years ago into the 21st century. New test methods have been introduced, and supplied are not only operation manuals printed in different languages, but also a complete range of lab ware, starter kit chemicals and reference standards is included - all suitably packed for global shipment by air. Now, identification of counterfeit medicines containing wrong, too little, or no ingredients at all can be performed instantly anywhere in the world. Results obtained by a set of physical and chemical screening tests must match the product label claims for, at least, drug identity and content. If they do not match or results are inconclusive, then the appropriate batches can be frozen for further investigation in a fully-fledged lab.
Simple Drug Quality Verification in Four Steps
 Verifying the quality of drugs using the GPHF-Minilab™ involves a four-level test scheme that employs very simple physical and chemical testing:
Verifying the quality of drugs using the GPHF-Minilab™ involves a four-level test scheme that employs very simple physical and chemical testing:
- A physical inspection scheme of dosage forms and associated packaging material for an early rejection of the more crudely presented counterfeits
- A quick check of the fill and total weight serves as an early indicator for the detection of false information related to the drug content
- A simple tablet and capsule disintegration test in order to verify label claims on enteric-coating and other modified-release systems
- Easy-to-use thin-layer chromatography as chemical test for a rapid verification of label claims regarding drug identity and content
Drug Quality Verification at Low Costs
GPHF-Minilabs contain the essential lab ware, chemicals, and a full range of control substances for reference purposes. Supplies include sufficient quantities in order to perform about 1000 assays while ensuring that the total material costs for one test run do not exceed three Euros.
A Complete Laboratory in One Suitcase
A heavy-duty flight case contains the essential components - a full range of glassware for sample extraction, preparation, pipetting and spotting, high performance chromatographic plates, developing and detection chambers, electronic pocket balance, UV lamps with different wavelengths, a hot plate and calliper rules. Even pens and pencils are included.
In addition, the flight case contains a manual with simple operation procedures and a collection of reference agents for up to 125 active pharmaceutical ingredients and some contaminants. Reducing sciences to working practice and richly illustrated, the manual reads more like a cooking recipe than a complex instruction booklet. The manuals method inventory is also available in French and Spanish. The suitcase is accompanied by a set of chemicals that allows the immediate use of Minilabs in any corner of the world.
Succesful Networking and Minilab Use
Priority disease programs frequently use GPHF-Minilabs to monitor the quality of essential medicines in malaria-, TB- and AIDS-endemic countries. Further multilateral support comes from the institutions and organisations of the United Nations family, such as the World Health Organisation (WHO), and beyond this, the Global Fund (GFATM). Bilateral support for the Minilab project work is mostly provided by programmes of the US Agency for International Development Cooperation (USAID), e.g. the Promoting the Quality of Medicines (PQM) programme currently implemented by the US Pharmacopeial Convention (USP). Over the years, in cooperation with the local authorities, the quality of medicines in various segments of the pharmaceutical supply chain has been repeatedly monitored in sub-Saharan Africa and Southeast Asia and, as a result, with support by the International Criminal Police organisation INTERPOL, millions of falsified malaria tablets without active ingredients have already been intercepted.
Within international development cooperation, additional support for the use of GPHF-Minilabs comes from governmental and non-governmental health projects, e.g. from Great Britain (Crown Agents), Switzerland (Swiss Tropical and Public Health Institute), Belgium (BTC/CTB) and Germany (GIZ). Non-governmental organisations as well as faith-based and university institutions, mostly from Germany complement government interventions to combat falsified and substandard medicines and use GPHF-Minilabs for their own health services, hospitals and research projects (DIFÄM, MMI Würzburg, Action Medeor, German Doctors, Tubingen University).

Other Minilab users are, for example, public drug procurement agencies and faith-based drug supply organisations in Nicaragua and Bolivia, medicines regulation authorities in Indonesia and Papua New Guinea, consumer protection institutions in India and Pakistan, diocesan hospitals in Ghana and Cameroon, scientific societies and business institutions in Laos and Thailand as well as the Bill and Melinda Gates Foundation (BMGF) in cooperation with the London School of Hygiene and Tropical Medicine (LSHTM) for rolling back malaria projects in East Africa.
More than 1000 Minilabs have been supplied to more than 100 countries out of which Tanzania, Uganda and Myanmar adopted the technology for post-marketing antimalarial drug quality monitoring already.